- Blog
- Chess Visualization Course Pdf
- Dw Drums Serial Number Decoder
- Fall Out Boy Discografia Torrent
- Peta Bali Lengkap Dengan Skala Dan Legenda
- Evang. Mba Abaraogu Onye Ga Bu Nwanne M
- Hdd Regenerator 2011 Crack
- Pvr Iptv Simple Client
- Case 235h Manual
- Kodi Skin Repo
- Download Game Tembak Tembakan Offline Pc
- Jetman Complete Download
- Din Oso 2768
- Verifications.io Database Download
- Facial Expressions References
- School Ein Georgia Tech
- Contoh Kata Pengantar Laporan Pkl
- Classic Wow Gm Commands
- Hindi Voice Typing Online
- How use fireshot chrome ecyension
- Fortnite direct downlaod link for android
- Statgraphics centurion xvii numero de serie uva
- Change db from german to english simatic manager
- 1964 gibson es 125 for sale
- Devi bhagavatam tamil pdf
- Handbrake choppy video audio
- Es file explorer pro vs es file explorer premium
- Runa laila pakistani urdu songs
- Internal combustion 4 stroke gas
- Photoshop 2013 torrent
- Delete all soundpools magix music maker
This means that, in order to burn completely 1 kg of fuel, we need 14.7 kg of air. For a gasoline (petrol) engine, the stoichiometric air fuel ratio is around 14.7:1. The ideal (theoretical) air fuel ratio, for a complete combustion, is called stoichiometric air fuel ratio. For example, if we have a mixture of methane and air which has the air fuel ratio of 17.5, it means that in the mixture we have 17.5 kg of air and 1 kg of methane. A complete combustion takes place when all the fuel is burned, in the exhaust gas there will be no quantities of unburnt fuel.Īir fuel ratio is defined as the ratio of air and fuel of a mixture prepared for combustion. To guarantee the combustion process, certain quantities of fuel and air need to be supplied in the combustion chamber.

Thermal engines use fuel and oxygen (from air) to produce energy through combustion. Impact of air fuel ratio on engine emissions.How stoichiometric air fuel ratio is calculated.But the overall concept is still the same. Some cars may have slight differences in this process, like with the number of cylinders it has. Either that or it would gradually damage the components of the engine.

If there was a problem with any one of these strokes then it would prevent the whole combustion cycle from happening. The cycle keeps repeating over and over again as you put your foot on the gas pedal to accelerate the vehicle. The engine has now gone through one rotation of the four-stroke combustion cycle. All of the exhaust that was created in the cylinder starts to leave through the exhaust valve and comes out of the car’s tailpipe. This is known as the power or combustion stroke.Īfter the explosion, the piston falls back down to the bottom and causes the exhaust valve to open up. There is a mini-explosion inside of the cylinder where the gasoline charge is still active. Once it does, a spark is discharged by the spark plug which causes the gasoline to ignite. The piston eventually makes it back to the top after compressing the air and gasoline mixture. Just a small drop of gasoline mixed together with air will create the stroke.Īfter that, the piston moves upwards and compresses the mixture of air and gasoline to give it a more powerful effect. There doesn’t have to be a lot of gasoline for this to work either. The intake stroke occurs when the gasoline mixes with the air. From there, the piston allows gasoline and air to get into the engine from the cylinder. The piston moves from the top to the bottom once the intake valve opens up. What happens is there’s a connecting rod that connects the piston to the crankshaft. The intake stroke is the first part of the internal combustion process and is basically the aspiration or breathing of the engine. See Also: What Happens If Your Put Gasoline in a Diesel Engine? #1 – Intake Stroke It is important to understand the term Otto Cycle because it is different than the combustion cycle that diesel engines used known as the “Diesel Cycle.” This cycle is also a four-stroke process, but the details of how each process works are different than the Otto Cycle.īelow are the four unique processes for combustion to take place in a typical gasoline engine. Each step in the process is named with the word stroke in it intake stroke, compression stroke, power stroke, and exhaust stroke. A German engineer named Nikolaus Otto was the first person to invent and patent a four-stroke gas engine.

The four-stroke engine process is also known as the Otto Cycle.
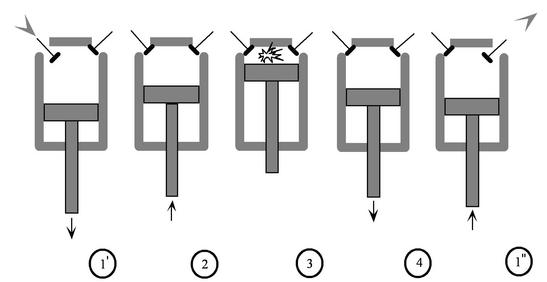
Related: Diesel Engine vs Gasoline Engine Keep reading to understand (and see) just how a gas engine works that’s found in most vehicles on the road today. Virtually every car that you see on the road uses a four-stroke combustion cycle to create motion from gasoline. When you are able to harness the expanding gas that comes from this process, then you have created the core of an internal combustion engine.įrom there, the energy from this gas is converted into motion. Internal combustion is based on the idea that you can create lots of energy when you burn gasoline in a small enclosed area.
